|
����RESULTS
����In three groups��drug-applied eyes exhibited lower evans blue extravasation �ۣ�6.50��2.34����g��ml��g for CKF����6.76��2.32����g��ml��g KF and ��11.22��3.24����g��ml��g DM��than those of the relevant vehicle-applied eyes�ۣ�14.94��4.49����g��ml��g����10.46��5.15����g��ml��g and ��13.74��3��87����g��ml��g��respectively�ݣ�The statistical differences between drug-and vehicle-treated eyes were significant in CKF ��P��0.0007�� and KF ��P��0.043�� groups����See Tab��1��
Tab��1 Evans blue��EB��extravasation in ocular tissues
| Drugs |
EB extravasation in eyes ����g��ml��g�� |
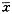 ��s ��s |
P value |
| 1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
9 |
| CKF |
4.38 |
4.68 |
7.20 |
5.15 |
6.27 |
7.08 |
11.90 |
7.31 |
4.58 |
6.50��2.34 |
�� |
| Veh |
23.97 |
7.32 |
14.26 |
11.95 |
16.83 |
15.98 |
16.89 |
14.29 |
13.02 |
14.94��4.49 |
0.0007 |
| KF |
4.46 |
4.75 |
4.56 |
4.95 |
10.44 |
7.96 |
6.33 |
7.54 |
9.88 |
6.76��2.32 |
�� |
| Veh |
5.50 |
3.65 |
9.09 |
19.06 |
15.27 |
6.81 |
7.74 |
12.35 |
14.72 |
10.46��5.15 |
0.043 |
| DM |
8.21 |
6.16 |
13.45 |
9.62 |
12.53 |
10.48 |
17.43 |
12.24 |
10.82 |
11.22��3.24 |
�� |
| Veh |
6.28 |
17.57 |
15.73 |
11.20 |
19.38 |
14.90 |
11.63 |
14.34 |
12.67 |
13.74��3.87 |
0.16 |
CKF��Compound ketotifen eye drops��KF��0.1�� Ketotifen eye drops��DM��0.0005�� Dexamethasone eye drops��Veh��Vehicle
����EB leakage significantly decreased by 53.98����15.57�� in CKF treated-eyes��when compared with either KF��27.91����28.36����P��0.028��or DM��11.90����34.16����P��0.004�� treated eyes��The differences were statistically significant����See Tab��2��
Tab��2 Inhibition rates of the drugs on evans blue leakage in eyes������
| �� |
Inhibition of EB extravasation in eyes |
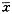 ��s ��s |
| 1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
9 |
| CKF |
81.37 |
36.06 |
49.51 |
56.90 |
62.74 |
55.69 |
29.54 |
48.84 |
64.82 |
53.98��15.57 |
| KF |
18.91 |
��30.14 |
49.83 |
74.03 |
31.63 |
16.89 |
18.22 |
38.95 |
32.88 |
27.91��28.36 |
| DM |
��30.73 |
64.94 |
14.49 |
14.15 |
35.34 |
29.62 |
��49.87 |
14.64 |
14.52 |
11.90��34.16 |
������CKF had a significantly high inhibition rate��when compared with either KF ��P��0.028��or DM��P��0.004����
����DISCUSSION
����Allergic conjunctivitis is a response resulting from a reaction of allergen with IgE antibodies on mast cells in the conjunctival stroma��This reaction triggers a cascade of events resulting in the degranulation of mast cells��and releasing chemical mediators��e��g����histamine��prostaglandins and leukotrienes�� into the conjunctival stroma and tears��These mediators cause vasodilation and increasing in vascular permeability��which induce ocular itching��burning��conjunctival injection��chemosis and limbal hyperemia����4��5��
����Corticosteroids��histamine H1-receptor antagonists��mast cell stabilizers and non-steroidal anti-inflammatory drugs��which exert antiallergenic effects by different pharmacological mechanism��have been used to treat anaphylactic ocular disorders��4��5����
����Ketotifen is the second generation histamine H1-receptor antagonist and also possesses stabilizing mast cell activity��which prevents the mast cell degranulation and chemical mediators release in the conjunctiva��and reduces the ocular symptoms and signs��1��6����Hagihara��7�� has clinically evaluated oral ketotifen for the treatment of 22 cases with efficacy rate of 80����No serious drug side effect was found in his study��Mikuni��8�� determined quantitatively the therapeutic effect of 0.08�� ketotifen eye drops in 10 volunteers suffering from seasonal cedar pollinosis provocated during a quiescent stage��The efficacy rate of the preparation was found to be 80����
����It is well-known that corticosteroids have a pronounced antianaphylactic action��The application of topical corticosteroids is now an indispensable modality in the treatment of severe ocular allergic conditions��Unfortunately��topical ocular corticosteroids may cause serious adverse effects��such as glaucoma��cataract formation��diminished epithelial wound healing��and the risk of ocular infection��which make long-term corticosteroid use invadisable��However��there was evidence that an administration of micro-dexamethasone could not only offer its desired pharmacological activity��but also minimize the adverse effects��9����
����In order to avoid the corticosteroid side effects and retain the antiallergenic effect��we conducted micro-dexamethasone and prepared topical ophthalmic formulation with 0.1�� ketotifen and 0.0005�� dexamethasone in this study��The micro-dexamethasone-applied eyes revealed lower EB extravasation than that of the control eyes��although no statistically significant difference was revealed between the two treated groups��The combination of 0.1�� ketotifen and 0.0005�� dexamethasone were a synergic effect on ocular anaphylaxis��inhibiting remarkably ocular evans blue extravasation in the model of acute allergic conjunctivitis with an inhibitory ratio of 53.98����which was considerably higher than that of each drug used alone��27.91�� and 11.90�� for 0.1�� ketotifen and 0.0005�� dexamethasone��respectively����The differences were statistically significant��The results of this study suggested the potential of compound ketotifen eye drops in the treatment of allergic conjunctivitis��
����REFERENCES
����1��Grant SM��Goa KL��Fitton A��et al��Ketotifen��A review of its pharmacodynamic and pharmacokinetic properties��and therapeutic use in asthma and allergic disorders��Drugs��1990��40��3����412
����2��Calonge MC��Pastor JC��Herreras JM��et al��Pharmacologic modulation of vascular permeability in ocular allergy in the rat��Invest Ophthalmol Vis Sci��1990��31��1����176
����3��Trocme SD��Trocme MC��Bloch KJ��et al��Topically induced ocular anaphylaxis in rats immunized with egg albumin��Ophthalmic Res��1986��18��1����68
����4��Jaanus SD��Hegeman SL��Swanson MW��Antiallergy drugs and decongestants��In��Bartlett JD��Jaanus SD��eds��Clinical Ocular Pharmacology��3rd ed��Boston��Butterworth-Heinemann��1995��337-53
����5��Abelson MB��Sloan JE��Mooshian M��Basis of ocular allergy and mechanisms for successful management��Ophthalmoic Practice��1993��11��6����278
����6��Heyman SN��Karmeli F��Brezis M��et al��The effect of ketotifen on nitric oxide synthase activity��Br J Pharmacol��1997��120��8����1545
����7��Hagihara M��Sakashita M��Inoue A��Clinical evaluation of ketotifen ��zaditen�� of the treatment of allergic conjunctival disorders��Folia Ophthalmol Jpn��1988��39��392
����8��Mikuni L��Sato K��Togawa K��et al��A quantitative tear fluids determination of therapeutic efficacy for allergic conjunctivitis��Tokai J Exp Clin Med��1984��9��1����35
����9��Chen ZJ��Practical Ocular Pharmacology��Beijing��Scientific %26 Technological Press of China��1993��212-34
(�ո壺1999��09��15 �أ�2000��01��08) ��һҳ [1] [2] |
